The rapid large rise in blood glucose following consumption of high-GI food triggers a large insulin response and strongly inhibits glucagons release. For most foods, a good correlation exists between glucose and insulin responses, with high-GI foods eliciting large insulin responses [35], which trigger rapid uptake of nutrients by insulin-responsive tissues and suppress nutrient mobilization. Glucose uptake and glycogen synthesis in skeletal muscle and liver, and lipogenesis in adipose tissue, are increased. Simultaneously, gluconeogenesis and glucose out put by liver and lipolysis are suppressed.
Low-GI diets give a more stable diurnal profile, reducing postprandial hyperglycaemia and hyperinsulinaemia, and attenuating late postprandial rebounds in circulating free fatty acids, all factors that exacerbate various components of the metabolic syndrome [23].
B. Insulin resistance and insulin secretion
High circulating free fatty acids levels result in lipid accumulation in skeletal muscle and liver, causing insulin resistance in these normally insulin-responsive tissues [36,37], which reduces insulin-stimulated glycogen synthesis in skeletal muscle (the primary pathway for non-oxidative glucose disposal in normal subjects [38] and decrease the ability of insulin to suppress hepatic glucose production and output.
Insulin sensitivity may be negatively affected in the long term as low- carbohydrates, high-fat diets favor an increase of plasma circulating free fatty acids [39,40], which under usual dieting conditions is typically associated with many insulin-resistant states in humans [41,42]. Altered fatty acid metabolism contributes to insulin resistance because of alterations in the partitioning of fat between the adipocytes and muscle or liver [15]. Accumulation of fatty acid and fatty acid metabolites in these insulin-responsive tissues leads to acquired insulin signaling defects and insulin resistance resulting in a reduced glucose transport [43]. The later is thought to results from fatty-acid-induced alterations in upstream insulin signaling events, resulting in decreased GLUT 4 translocation to the plasma membrane. Increased level of intracellular fatty acid metabolites, such as diacylglycerol, fatty acyl CoA's, or ceramides activates a serine/threonine kinase cascade, possibly initiated by protein kinase CӨ. The latter leads to a non-desired phosphorylation of serine/threonine sites on insulin receptor substrates, which then fail to associate with or to activate PI 3-kinase, resulting in decreased activation of glucose transport and other downstream events [15].
Fig. 2 Regulation of insulin secretion by glucose and lipids (adapted by Brand et al. (2004). Free Radic. Biol. Med.).
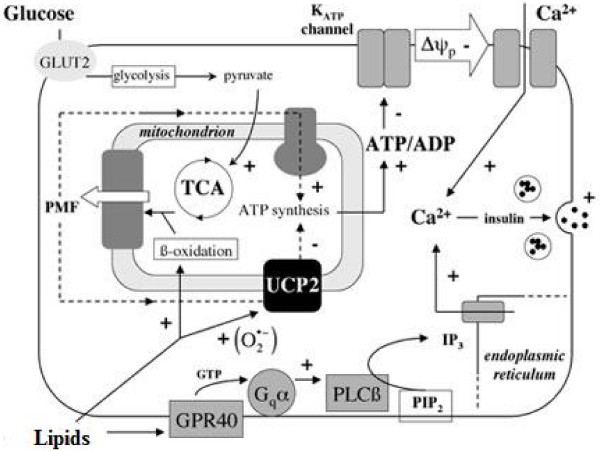
Description: Insulin secretion is influenced by plasma glucose and lipids that activate on one hand glycolysis and production of pyruvate, and on the other hand β-oxidation and ATP synthesis.
References



